CytoNiche’s 3D FloTrix™ platform won the Emerging Bioprocessing Supplier – Downstream award at ABEA 2026, recognising scalable 3D cell therapy manufacturing.
SINGAPORE, March 18, 2026 /EINPresswire.com/ — At a pivotal moment for the Asia-Pacific regenerative medicine sector, CytoNiche’s 3D FloTrix™ technology has been named the winner of the Emerging Bioprocessing Supplier Award: Downstream Processing at the Asia Pacific Biopharma Excellence Awards (ABEA) 2026. The prestigious ceremony, held at the Sands Expo & Convention Centre, brought together the architects of tomorrow’s healthcare to recognise innovations that bridge the gap between laboratory breakthroughs and commercial-scale reality.
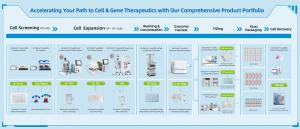
The ABEA awards serve as a regional benchmark, celebrating outstanding achievements in bioprocessing, logistics, and clinical trials. This specific accolade highlights a key shift in the industry: the transition from traditional 2D manual cultures to automated, scalable 3D microcarrier suspension systems designed to support mass manufacturing of cell therapies, including mesenchymal stem cells (MSCs), exosomes, and other advanced therapy products.
Advancing the Frontiers of Regenerative Medicine
The recognition in the “Emerging Bioprocessing Supplier” category underscores CytoNiche’s rapid trajectory towards solving complex challenges in the purification and processing of advanced therapies. By driving the industry’s move away from labour-intensive 2D manual cultures toward closed-system, automated 3D microcarrier suspension systems, CytoNiche provides a comprehensive end-to-end solution for Cell and Gene Therapy (CGT) manufacturers. This integration of sophisticated downstream capabilities ensures that life-saving treatments can be produced with significantly greater efficiency and cost-effectiveness.
“This award is more than a trophy; it is a validation of our vision to industrialise cell manufacturing. At the heart of this success is our 3D TableTrix™ Microcarrier—a porous, biodegradable scaffold specifically engineered to harmonise high-yield cell expansion with seamless downstream harvesting,” said Dr. Yan Xiaojun, Co-founder & CTO of CytoNiche Biotech. “Being recognised by our peers in the Asia-Pacific region motivates us to continue pushing the boundaries of what is possible in bioprocessing—ensuring these life-saving treatments are both scalable and accessible.”
Scaling the Future of Biopharma
As the industry pivots towards closed-system, automated manufacturing, the focus has shifted from mere feasibility to sustainable, high-volume production. The ABEA 2026 trophy and certificate of excellence symbolise CytoNiche’s trajectory as a global leader and its unwavering commitment to advancing global access to cell technologies through disruptive and scalable manufacturing innovations.
By championing the “Cell Architects” philosophy, CytoNiche is not just providing tools but is fundamentally redesigning the manufacturing blueprint. The company continues to lead the charge in transitioning the field towards intelligent, automated 3D platforms. These specialised systems—optimised for Mesenchymal Stem Cells (MSCs), NK cells, exosomes, and viral vector production, etc.—ensure that as the demand for regenerative medicine grows, the infrastructure to support it is robust, reproducible, and ready for the global stage.
About CytoNiche
Founded in 2018, CytoNiche Biotech is a pioneering provider of 3D cell mass production systems for therapeutic applications. Built on its proprietary dissolvable microcarrier technology, CytoNiche’s 3D Cell Intelligent Manufacturing system has supported the regulatory approval of China’s first stem cell drug, Amimestrocel Injection. Its flagship 3D FloTrix™ platform facilitates a seamless transition from antiquated 2D manual cultures to scalable, high-fidelity, closed-system 3D suspension environments and enables cost-effective, large-scale manufacturing of cell and gene therapies. The platform features GMP-compliant dissolvable microcarriers, serum-free media for mesenchymal stromal/stem cells, and a complete suite of automated bioreactors and processing systems and single-use, closed-loop consumables. CytoNiche supports every stage of the cell therapy pipeline—from cell expansion to harvesting, processing, and final fill.
For more information, please visit: https://en.cytoniche.com
About the Asia Pacific Biopharma Excellence Awards (ABEA)
The Asia-Pacific Biopharma Excellence Awards (ABEA) are the region’s most prestigious benchmark for operational and technological mastery in the life sciences. ABEA serves as a critical barometer for industry evolution, identifying organisations that successfully bridge the gap between complex R&D and commercial-scale accessibility. By honouring achievements across bioprocessing, logistics, and clinical trials, the awards recognise “trailblazers” who de-risk the path to market for next-generation medicines, inspiring innovators to build a more resilient, efficient, and patient-centric global healthcare infrastructure.
Winnie Ng
Cytoniche Biotech Pte. Ltd.
marketing@cytoniche.com
Visit us on social media:
LinkedIn
Facebook
YouTube
Legal Disclaimer:
EIN Presswire provides this news content “as is” without warranty of any kind. We do not accept any responsibility or liability
for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this
article. If you have any complaints or copyright issues related to this article, kindly contact the author above.
![]() Topic / Science, Topic / Healthcare & Pharmaceuticals Industry, Topic / Technology, Topic / Media, Advertising & PR, Topic / Manufacturing, Country / Indonesia, Country / United States
Topic / Science, Topic / Healthcare & Pharmaceuticals Industry, Topic / Technology, Topic / Media, Advertising & PR, Topic / Manufacturing, Country / Indonesia, Country / United States




Be First to Comment